Self-Disseminating Vaccines
Outbreaks of lethal viruses in humans including Ebola, Nipah, and SARS have directed global attention to bats as among the most important and least understood viral reservoirs. Impacts of bat-associated zoonoses are currently mitigated reactively, by treating “spillover hosts” (humans and domestic animals) after viruses emerge. However, this approach indefinitely prolongs the risk of spillover and epidemic spread, allowing enormous health and economic costs to accumulate. Alternatively, vaccinating reservoir hosts can reduce spillover by eliminating the source of infection. Currently, operational challenges to scalability currently limit practical feasibility in most wildlife, including bats. Vaccines that autonomously spread from vaccinated to susceptible hosts (self-disseminating vaccines) are a potentially game-changing solution. Using vampire bat rabies, we are exploring two candidate strategies using recombinant virally-vectored vaccines: transferable vaccines which use a Raccoonpox vector that spreads only from treated bats and transmissible vaccines that will use a recombinant betaherpesvirus to spread infectiously over multiple generations. We seek to appraise a new reservoir-targeted vaccination paradigm that could bring elimination of bat viruses into sight for the first time.
Key publications
Longitudinal deep sequencing informs vector selection and future deployment strategies for transmissible vaccines. PLoS Biology (2022)
Self-spreading vaccines – base policy on evidence. Science (2022)
Epidemiology and biology of a herpesvirus in rabies endemic vampire bat populations. Nature Communications (2020)
Fluorescent biomarkers demonstrate prospects for self-spreading vaccines to control disease transmission in wild bats. Nature Ecology & Evolution (2019)
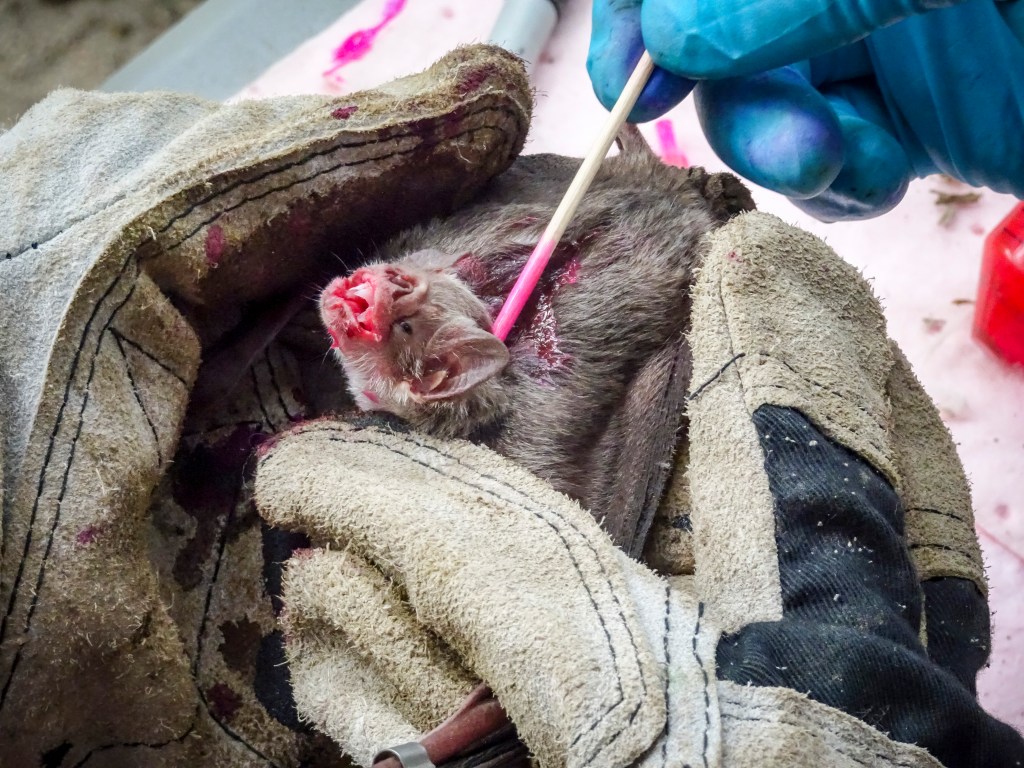
Forecasting rabies
Anticipating how epidemics spread across landscapes requires understanding host dispersal events that are notoriously difficult to measure in wildlife. Our work has used spatiotemporal models, viral phylodynamics and host population genetics to anticipate future vampire bat rabies outbreaks in humans and domestic animals in Latin America. New work funded by the Wellcome Trust is evaluating how landscape characteristics and spatiotemporal machine learning can improve the accuracy and precision of predictions with the ultimate aim of generating a real-time early warning system that could enable anticipatory vaccination of humans and livestock.
Key publications
Phylodynamics reveals extinction-recolonization dynamics underpin apparently endemic vampire bat rabies in Costa Rica. Proceedings of the Royal Society B (2019)
Host-pathogen evolutionary signatures reveal dynamics and future invasions of vampire bat rabies. Proceedings of the National Academy of Sciences of the USA (2016)
Spatial expansions and travelling waves of rabies in vampire bats. Proceedings of the Royal Society B (2016)
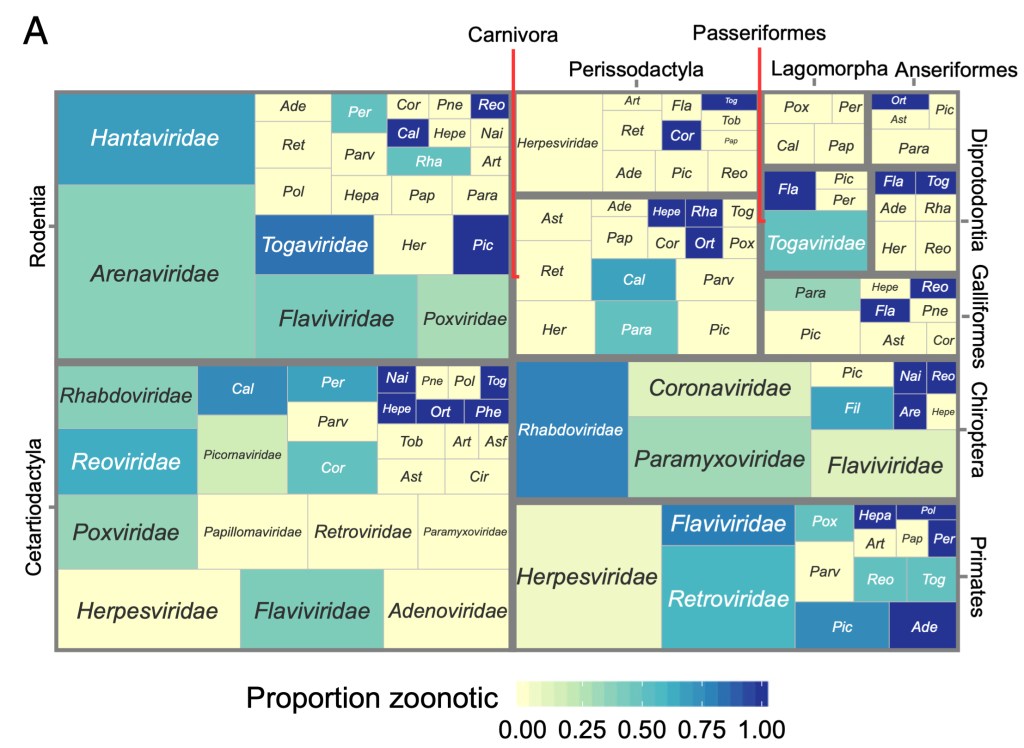
Viral origins and risk
Quantitative syntheses that identify general patterns in emerging infectious diseases can challenge conventional wisdoms, define new research directions, and guide management decisions. Using comparative multi-host, multi-virus datasets, we showed that viral zoonotic risk is relatively homogenous among orders of mammals and birds, suggesting that efforts to predict unrealized zoonoses might focus on identifying viral traits which predispose zoonotic transmission. Using large genomic and ecological datasets, we also demonstrated that the animal reservoirs and the existence and identity of arthropod vectors can be predicted directly from viral genome sequences via machine learning. Models such as these, which capitalize on the proliferation of low-cost genomic sequencing, can narrow the time lag between virus discovery and targeted research, surveillance, and management. Current projects are exploring how pairing machine learning with other kinds of ‘omics data can enhance our understanding of viral ecology and host range.
Key publications
Identifying and prioritizing potential human-infecting viruses from their genome sequences. PLoS Biology (2021)
Viral zoonotic risk is homogenous among taxonomic orders of mammalian and avian reservoir hosts. Proceedings of the National Academy of Sciences (2020)
Predicting Reservoir Hosts and Arthropod Vectors from Evolutionary Signatures in RNA Virus Genomes. Science (2018)
Ecological consequences of vampire bat culls
Disease control interventions in wildlife can lead to unexpected changes in animal ecology (e.g., dispersal) and demography (e.g., birth or survival rates) which have counterproductive effects on human and animal health. Vampire bat rabies is the only bat-associated zoonosis for which bats are routinely culled for disease control. While culls are likely to alleviate bat bites on humans and domestic animals, our field studies have shown that the core epidemiological assumptions underlying culling as a disease control policy are not supported by data. Moreover, mathematical models suggested that if culls increase bat dispersal, this could enhance rather than dampen rabies transmission. New research funded by an NSF/BBSRC Ecology and Evolution of Infectious Diseases Grant will test how culls alter bat demography and dispersal. Simultaneously, work funded by the Wellcome Trust is filling knowledge gaps in the biology of rabies using experimental infections in captive bats and new field monitoring techniques which will let us optimize alternative control strategies.
Key publications
Resolving the roles of immunity, pathogenesis and immigration for rabies persistence in vampire bats. Proceedings of the National Academy of Sciences of the USA (2013)
Ecological and anthropogenic drivers of rabies exposure in vampire bats: implications for transmission and control. Proceedings of the Royal Society B (2012)

Other curiosities
From time to time we discover interesting microbes using metagenomic sequencing or have a question that can’t be answered with rabies or any single virus, but require a whole pathogen community approach.
Key publications
Diversification of mammalian deltaviruses by host shifting. Proceedings of the National Academy of Sciences of the USA (2021)
Demographic and environmental drivers of metagenomic viral diversity in vampire bats Molecular Ecology (2020)
